We are all motivated to eat. This is a basic function of survival. In fact, our brains are equipped with an ancient motivational system that ensures we are driven subconsciously to hunt and gather (“go and get”) food, particularly calorie-dense foods. This ancient system can lead to overeating and weight gain in our modern food environment.
Earlier modules have described overweight and obesity as a real, genetically influenced, brain-centred, progressive, chronic disease. Earlier modules have also shown that effective treatment is not limited to willpower or lifestyle advice alone. Behavioural treatment remains a foundational treatment pillar. Obesity medications are now an important second pillar, and bariatric surgery remains an important third pillar. The focus of this module is obesity medication.
Today, safe and effective obesity medications play a central role in obesity care.
In Canada, long-term pharmacotherapy options now include liraglutide, naltrexone/bupropion, orlistat, semaglutide, tirzepatide, and setmelanotide for specific rare genetic obesities. These medications are used alongside health-behaviour change and should be individualized based on expected benefit, tolerability, contraindications, route of administration, cost, access, and patient preference.
Medical therapy has advanced substantially, and surgery remains an important treatment option, especially for severe obesity.
The field has changed meaningfully since this module was first written. Newer incretin-based therapies have produced greater average weight loss and broader health benefits than earlier medications, and semaglutide has also shown cardiovascular benefit in selected populations. At the same time, bariatric surgery remains a highly effective treatment option. Modern obesity care should therefore present medication and surgery as complementary tools within chronic disease treatment, not as competing ideologies.

In Canada, obesity surgery includes the sleeve gastrectomy, the gastric bypass or the duodenal switch. You may think that the weight loss effects of these procedures take place at the level of the abdomen, however, perhaps surprisingly, the majority of the weight loss effects of these procedures is accomplished via a strong influence on all three layers of the appetite system as described here in the case of gastric bypass.
Obesity medications work in different ways. Some, such as orlistat, work mainly in the gut. Others act more directly on the biology of appetite, hunger, fullness, and food reward. This includes naltrexone/bupropion, liraglutide 3.0 mg, semaglutide 2.4 mg, and tirzepatide. The newer medications have expanded what is possible with medical obesity treatment and have changed the treatment landscape substantially since earlier versions of this module were written.

Liraglutide 3.0 mg and semaglutide 2.4 mg are GLP-1–based medications that increase fullness and reduce hunger. Tirzepatide works through both GIP and GLP-1 pathways and also increases fullness while decreasing hunger and calorie intake. In practical terms, these medications help quiet the biological pressures that defend against weight loss and push appetite upward.

The central role of WANTING has been established in the WANTING module.
WANTING drives overeating and can make weight management much harder. Naltrexone/bupropion acts on appetite and food reward pathways, and newer incretin-based medications such as semaglutide and tirzepatide also appear to reduce hunger, food preoccupation, and cravings in many patients. In other words, these medications do not simply depend on willpower — they help change the biological drive to eat.

One way to understand this effect is to look at appetite, craving, and eating-control measures used in clinical trials. Semaglutide has been shown to improve control of eating, reduce hunger, and reduce food cravings. These effects help explain why newer obesity medications can produce substantially greater and more sustained weight loss than older therapies.
The key message is that obesity medications can influence the brain systems involved in fullness, hunger, and food reward. This is why medication can be a powerful addition to behavioural treatment: it can help reduce the biological and psychological pull toward overeating while the patient continues to build skills, routines, and restraint.
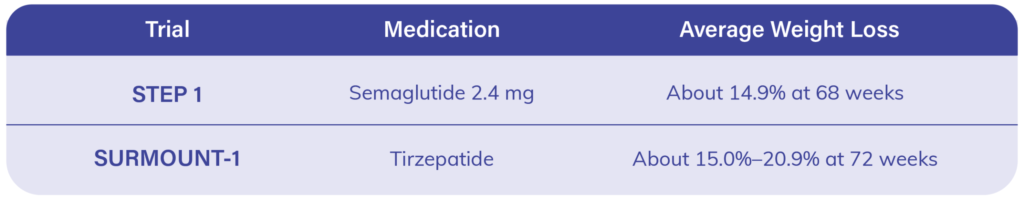
Clinical trials are conducted to determine the safety, tolerability, and effectiveness of obesity medications. Since this module was first written, the expected outcomes with medication have changed meaningfully. Earlier medications helped many people achieve modest-to-moderate weight loss. Newer incretin-based medications have moved average outcomes into a new range and have reshaped expectations for what medical obesity treatment can achieve.
In the STEP 1 trial, semaglutide 2.4 mg, combined with lifestyle intervention, resulted in an average weight loss of approximately 14.9% at 68 weeks.
In SURMOUNT-1, tirzepatide produced average weight loss ranging from about 15.0% to 20.9% at 72 weeks, depending on dose. These results show how much the field has advanced beyond the earlier generation of obesity medications.
Not every person responds to every medication in the same way. Some people notice an early and powerful reduction in appetite, cravings, and food noise, while others have a smaller response or find a given medication difficult to tolerate. A limited response to one medication does not mean that another medication will not work well for the same person. For this reason, obesity medication treatment should be individualized, monitored, and adjusted over time.
The most important practical point is that modern obesity medications can produce clinically meaningful, sustained weight loss in many patients when combined with behavioural treatment. The degree of benefit varies, but the overall trend is clear: newer therapies have increased the likelihood of achieving larger weight reductions than were typically seen with earlier medications.
Treatment with obesity medications is also associated with improvements in weight-related health conditions such as blood pressure, blood sugar, cholesterol, physical function, and quality of life. In selected populations, newer medications such as semaglutide have also shown cardiovascular benefit beyond weight loss alone.
There are now several obesity medications approved in Canada. In routine adult obesity care, commonly used options include orlistat, naltrexone/bupropion, liraglutide 3.0 mg, semaglutide 2.4 mg, and tirzepatide. A more specialized medication, setmelanotide, is also approved for certain rare genetic obesities. Each medication has a different balance of benefits, risks, side effects, contraindications, route of administration, and access considerations.
All obesity medications should be reviewed carefully with a prescribing clinician. Tolerability varies by medication and by patient. Some people do very well with one medication and not another, and side effects, contraindications, cost, and insurance coverage often influence which option is most realistic and sustainable.
Which medication should be considered first depends on the individual. Exact response cannot be known in advance, but treatment choice can still be guided by clinical priorities, contraindications, co-existing health conditions, treatment goals, patient preference, affordability, and access. In other words, obesity medication selection is not random — it is individualized.
INDICATIONS
In Canadian obesity care, pharmacotherapy should generally be considered for adults with: A BMI of 30 kg/m² or above A BMI of 27 kg/m² or above in the presence of at least one adiposity-related complication such as hypertension, type 2 diabetes, dyslipidemia, obstructive sleep apnea, osteoarthritis, or other clinically important weight-related health problems.
CONTRAINDICATIONS
Each medication has its own contraindications, cautions, and monitoring requirements. These must be reviewed carefully with the prescribing clinician, since the safest and most appropriate choice depends on the individual medication and the patient’s medical history, concurrent medications, and treatment goals.
You have learned that many people living with overweight or obesity have internalized shame, blame, and the false idea that this condition is simply a matter of willpower. A more accurate and medically grounded view is that obesity is a complex, chronic, biologically regulated disease that is influenced by genetics, physiology, environment, and life experience. In that framework, medication is not a shortcut or a failure — it is one evidence-based treatment option for a real chronic disease. The decision to use medication remains yours, and the role of the clinician is to explain the potential benefits, risks, alternatives, and appropriateness of treatment for your situation.
This next point is important. Obesity medications work best when they are part of comprehensive long-term care. That care may include behavioural treatment, nutrition support, physical activity, sleep optimization, and psychological support where appropriate. Medication should not be framed as competing with behavioural care; rather, it can make behavioural treatment more effective by reducing hunger, cravings, and the biological pressure to regain weight.
When discussing who should consider obesity medication, it is helpful to think in terms of indications, contraindications, and personalization. Indications help identify who may benefit from treatment. Contraindications identify situations where a medication should not be used. Personalization helps match the right treatment to the right patient based on goals, health conditions, risks, preferences, and access.
The reasoning is not simply that weight is higher, but that excess adiposity is affecting health, function, quality of life, or future risk. In practice, medication is used as part of chronic weight management alongside health-behaviour change and long-term follow-up. Exact product indications vary somewhat by medication, but this is the broad modern Canadian framework.
Which medication should be considered first depends on the individual. Exact response cannot be known in advance, but treatment choice can still be guided by clinical priorities, contraindications, co-existing health conditions, treatment goals, patient preference, affordability, and access. In other words, obesity medication selection is not random — it is individualized.
Behavioural treatment remains a foundational pillar of obesity care, but modern obesity management is best approached as chronic disease care rather than a step-by-step test of willpower. Obesity medication can be considered at different points in treatment depending on health status, treatment goals, response to behavioural care, and patient preference. When considering medication use, the following four options can still be helpful ways to think about timing.
OPTION 1
Begin with behavioural treatment alone and do not use obesity medication at this time. This may be reasonable for some individuals, particularly if health goals are being met, weight-related complications are limited, or the person prefers to begin without medication. Behavioural treatment alone can absolutely be effective for some people. At the same time, modern obesity care recognizes that medication does not need to be reserved only for last-resort use if additional help is needed.
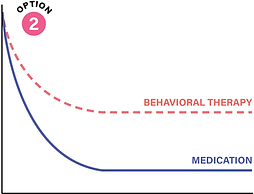
OPTION 2
Start medication at the same time as behavioural treatment begins.
This is now a very reasonable and often appropriate option, especially when earlier improvement in health, appetite control, weight-related complications, or quality of life is important. In current practice, obesity medications are recommended in conjunction with health behaviour changes, not as a replacement for them.
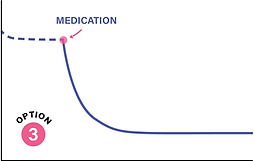
OPTION 3
Add medication after behavioural treatment has begun if progress is limited, weight-related complications remain active, or appetite biology continues to make change very difficult. This is also a common and appropriate pathway. In many patients, medication can reduce hunger, cravings, and food noise enough to make behavioural strategies easier to carry out and sustain.
A limited response to behavioural care alone should not be interpreted as failure; it may simply signal that additional treatment support is needed.
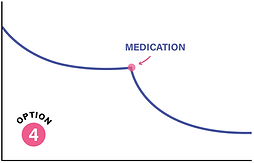
OPTION 4
Add medication when behavioural treatment alone has brought someone to their BEST WEIGHT, but health, function, or quality of life are still not where they need to be. Best weight is not the lowest possible weight — it is the weight a person can achieve and maintain while living their healthiest and happiest life. If that best weight still leaves meaningful adiposity-related complications, impaired function, or reduced quality of life, medication may help move treatment further in a helpful direction.
The timing of medication initiation should be discussed with a qualified clinician, but the decision should be individualized and made through shared decision-making. Once started, obesity medication should generally be titrated as needed and tolerated, continued long term if helpful, and reassessed over time based on treatment goals, health outcomes, tolerability, and patient preference.
Initiating obesity medication begins with a careful discussion of how the medication works, what benefits are being targeted, what side effects or contraindications need to be reviewed, and whether the treatment fits the patient’s goals, preferences, and circumstances. For many modern medications, treatment starts at a lower dose and is increased gradually to improve tolerability. For example, semaglutide is typically escalated stepwise to a maintenance dose, and tirzepatide is also started at a low weekly dose and increased gradually as needed and tolerated.
Because obesity is a chronic disease, obesity medication should usually be framed as part of long-term treatment rather than a short-term fix. When effective and tolerated, pharmacotherapy is generally continued long term to help maintain benefits and reduce the risk of weight regain or regression of health improvements. Treatment can still be reassessed over time if goals change, if side effects become limiting, or if access and affordability become major barriers.
After initiating obesity medication, follow-up should focus on more than the number on the scale. Assessment should include appetite response, hunger and craving control, tolerability, adherence, weight or waist-related progress if the patient wishes to track it, and improvement in obesity-related health complications, physical function, and quality of life. Treatment goals should be individualized and based on what matters most to the patient, not weight alone.
This is important. Obesity medication should no longer be viewed as care available only through a narrow group of specialists. Obesity treatment can and should increasingly be delivered within routine evidence-based medical care, including primary care. At the same time, some patients may still benefit from referral to clinicians or programs with additional obesity expertise, especially when treatment is complex, when multiple health conditions are involved, or when access and coverage are difficult to navigate.
Weight bias and outdated eat-less, move-more thinking still create barriers, so some patients may still need to advocate for evidence-based care. Patients should also use only authorized, prescribed medications and avoid unauthorized, counterfeit, or misleadingly marketed GLP-1 products sold online or through unofficial channels.
Access to obesity treatment is improving, but it still depends on local systems, clinician experience, and medication coverage, so finding the right care may require persistence and shared decision-making.
